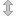Pretium iustum est:
The Price is Right?

Figure 1: “Melinda Townsend-Breslin holds a photo [of her mother and her]…in the parking lot of a favorite thrift store in 2013 a year before her death.”
"Medical Bills Linger, Long After Cancer Treatment Ends." NPR. NPR, 27 Mar. 2015. Web. 28 Jan. 2016.
People fight battles in hopes of finding peace. Fifty-eight-year-old MaryLou Townsend, a pancreatic cancer patient, approached her will to fight following her diagnosis in much the same way. However, somewhere between the frantic trips to the OR and frequent chemotherapy treatments, the bills piled up. And while the insurance company covered charges in the plan, the plan did not cover everything. Amanda Aronczyk, writer and reporter at National Public Radio, recounts the family’s story of how finances affected MaryLou’s care. During her treatment, Townsend suffered from blood clots and her body did not respond to the standard medication. The alternative, outpatient medication ended up costing over $1000 a month and “had to be paid out of pocket” (Aronczyk). As a result, the financial toll took more out of Townsend than the physical toll from her cancer. After Townsend’s death, her husband and daughter were left with around $100,000 in medical debt, which they slowly worked toward paying off.
Millions of American families face the looming cloud of rising pharmaceutical drug prices that overshadow the benefits of treatment each year. The price of prescription drugs for terminal illnesses contributes largely to medical expenses that often leads to bankruptcy. In 49 out of 50 states, the leading cause of bankruptcy in 2014 was found to be medical debt (Stech). Pharmaceutical companies go unchecked as they set astronomical, arbitrary prices on medicines for diseases. Due to the Food and Drug Administration’s (FDA’s), a regulative body responsible for promoting and protecting the state of public health in the nation, lack of regulation over pharmaceutical drug prices, the corporations who market and sell the biomedical drugs make enormous profits through price spiking while their consumers are left scrambling to pay for them.
America’s regulation of health policy began in 1984 with the Hatch-Waxman Act, more formally known as the Drug Price Competition and Patent Term Restoration Act. Its purpose encompasses the establishment of cheaper manufactured generic drugs by the pharmaceutical industry and marks the first regulation on drug pricing. Clinical trials, studies, and research that are normally performed prior to FDA approval for new medications are no longer requirements for small-molecule drug equivalents with active properties similar to already licensed medications. The act decreases prices up to 80% (Grabowski 2157). This victory in price reduction is short-lived due to the patent period extension in response to the lack of innovation incentive for brand-name companies in the Hatch-Waxman Act.
The United States increased its patent term to 20 years from the filing date, or 17 years from the issue date in 1995. The revision stands as a response to the Hatch-Waxman Act and the World Trade Organization’s (WHO) standards on intellectual property. The patent period extension provides innovation incentive for pharmaceutical companies by eliminating competing drugs equivalents in the market for lower rates. This permits them to set prices as they wish. According to a study conducted by Ronald J. Vogel, PhD, patent protection grants pharmaceutical companies “a period of above-normal profits for a technically [commercial] …product (1204). Patents also act as a safeguard for biomedical companies, “because pharmaceuticals that are not derived from biotechnology can be imitated easily and inexpensively” (Vogel 1204). Instead the extension causes exclusivity resulting in higher prices for longer periods of time before generics hit the market.
The Center for Medicare & Medicaid Services (CMS) sets the standards for health insurance coverage and has insured Americans for over 50 years and insures over 100 million Americans today, making it the largest health care insurance provider in the country. Although the 2006 amendment aims to bridge the gap between consumers and out-of-pocket prescription payments, as of 2003 the “Medicare Prescription Drug, Improvement, and Modernization Act forbids Medicare [and the FDA] from negotiating drug prices” (Tefferi 997). The 2003 Act was not overturned when Medicare Part D was added as a part of the 2006 amendment. The overlap speaks to the contradiction of U.S. law on health care regulations. If neither the largest medical service provider in the country nor the standard regulatory body cannot negotiate drug prices, it grants pharmaceutical companies the leeway to sell drugs at the highest, consumable market value.
Turns out not all press is good press, with the controversies that have brought pharmaceutical price gouging to the forefront of the media in the past year. Turing Pharmaceuticals, in September 2015, was caught in a controversy over the increased the cost of Antiretroviral pills (AIDS drug) by more than 5000 percent that they authorized overnight. After much public scrutiny and national media coverage, the former CEO, stated that they would “mull over the decision to reduce prices,” but no doubt, intentionally failed to mention how (Harven). The poorly documented journey of how Pfizer reached its price tag proves the necessity for regulation and finally has U.S. Congress rallying for its citizens. With the recent massive media coverage given to Big Pharma exploiting their sick consumers such as the cases with Turing Pharmaceuticals and Pfizer, Congress has taken to run a hearing against former CEO of Turing Pharmaceutical, Martin Shkreli. In December of last year, Pfizer, one of the largest multinational, pharmaceutical corporations, set the price for a new breast cancer drug at $9850 per month. The House bill introduced by Rep. Elijah Cummings stands as proof that Congress can no longer debate this issue as if it is just another political debate on the extent of regulating a capitalist free market economy. In place of a petty political debate stands a battle for the welfare of America’s constituents and their fiscal and physical health.
One solution to this national issue is the United States House of Representatives Bill 3513 and the equivalent Senate Bill 2023, the Prescription Drug Affordability Act of 2015. Representative Elijah Cummings of Maryland, and Senator Bernie Sanders of Vermont. introduced the Prescription Drug Affordability Act on September 16, 2015 on September 10, 2015 in the United States House of Representatives and Senate, respectively. The bill has not been voted on yet in either the House or the Senate. It allows CMS (Center for Medicare & Medicaid Services) to intervene in the regulating of prescription drug affordability. The Act states that all drugs covered under Medicare Part D, which includes all inpatient treatments and some outpatient treatments, will be subject to price negotiations with drug manufacturers. And those manufacturers will then be required to issue prescription drug rebates to qualified low-income individuals. Thus resulting in the closure of the Medicare Part D coverage gap. The bill will amend the Federal Food, Drug, and Cosmetic Act to “allow the importation by individuals of prescription drugs from Canada, and potentially other countries” as well as to establish “conditions on the award of market exclusivity” with severe consequences to the manufacturers who break these laws (U.S. House). Meanwhile this Act, if passed, will modify the Federal Trade Commission Act to enforce restrictions on “anticompetitive patent settlements [called] ‘pay-for-delay’…[that] block generic drug competition” (H.R.).
While the Prescription Drug Affordability Act covers many essential parts to the issue of unchecked, astronomical prescription drug pricing, it leaves room for quite a few loopholes. For this reason the a policy from physicians at the Mayo Clinic, a nonprofit worldwide leader in medical care, research and education, came up with their recommendations to fight rising drug costs in their scholarly article, In Support of a Patient-Driven Initiative and Petition to Lower the High Price of Cancer Drugs. Doctors like Ayalew Tefferi, M.D., hematologist at the Mayo Clinic, claim that a “post-FDA approval review mechanism to propose a fair price for new treatments” will help reduce drug costs (Tefferi). The petition states that the direct negotiation of drug prices by Medicare should be established, similar to the Prescription Drug Affordability Act of 2015. In order to solve the issue of newer drugs on the market not improving quality of life, but only increasing life span by a matter of weeks or days, the petition advocates for allowing Patient-Centered Outcomes Research Institute, department created by the Affordable Care Act Initiatives to evaluate the specific benefits of new treatments before the drugs are approved by the FDA to hit the market. Again similar to the above congressional bill, the article suggests for the legalization of imported prescription drugs and legislation prevent the pay-for-delay avenue that prevents access to generic medications.
If the policy recommendations of H.R. Bill 3513 and the Mayo Clinic Petition are established in conjunction with each other, they contain the power to revolutionize prescription drug affordability and fight the rising drug costs with as much punch as pharmaceutical companies are countering with. The establishment of these laws creates a comprehensive protection against pharmaceutical companies’ astronomical prices and greed, with a barely-existent dent on their profit margin. The recommendations confront the source of the issue through covering the basis of international drug equivalents importation, price negotiations, and FDA review processes in terms of changing the way this country and all its residents approach drug treatment programs and paying for them. The bill and article encompass delay of generic drugs to the market, patent system reform, increasing competition for medications using the thus-far successful method of America’s European counterparts. The House Bill is still undergoing changes regarding its content, but has steady support many Democrats and fair number of Republican congressmen giving it a fighting chance with the long-term money-saving investment outweighing its minimal implementation costs. These recommendations are the face of the consumer-driven grassroots movement fighting Big Pharma and their skyscraper prices on life-saving medication. Such a significant commodity must be available without an “if-clause” attached the life-saving contract. The Prescription Drug Affordability Act of 2015 and Mayo Clinic’s Petition to Lower the High Price of Cancer Drugs possess the potential to rid the pharmaceutical market of its exclusivity from public acquisition.
The root cause behind price gouging and the resulting market exclusivity over pharmaceutical prescription drugs stems from a lack of regulation by the CMS and FDA. There is currently no possible way of negotiating with pharmaceutical companies, before their market price is set at whatever irrational number they choose. Both H.R. Bill 3513 and the Mayo Clinic Petition remedy this by giving CMS negotiating power over biomedical firms “on behalf of beneficiaries for drugs covered under Medicare Part D” (U.S. House). Negotiation periods cap at 180 days. Within that time a price should be set, and the medication should be well on its way to the shelves. Negotiations will be key, because Dr. Hagop Kantarjian, Chairman of the Leukemia Department at the University of Texas Medical Anderson Cancer Center and his colleagues state that disease “regression and prolongation of life are the goals [which should]… guide drug pricing” (“Cancer Drug in the United States: Justum Pretium—The Just Price” 3600). This will allow the standard to be set based off of the FDA approval process and prices will be based off of the best recommendations for patient care both financially and physically.
Having CMS negotiate medication rates fares logically, according to Hagop Kantarjian, M.D., and his colleagues at UT Medical Anderson Cancer Center, “given that elderly people suffer…disproportionately” from various terminal illnesses and Medicare is the leading insurance provider in the United States, not only for elderly people, but also as a whole (“Cancer Drug in the United States: Justum Pretium—The Just Price” 3600). Now although, this directly does not affect patients covered by private insurance companies, it has been seen that those private insurance companies follow the model of CMS in terms of various levels of coverage plans. Whether other insurance providers follow CMS’s guidelines because it is easier for them to meet the government standards on coverage, or they do so because it is the best for competition is not made clear. Regardless, the result is that all insurance providers in America receive standard prices on prescription medications and the effect leaves no patient untouched. Hence, CMS’s negotiations will benefit all American residents no matter the source of their drug coverage plans. Healthcare is a trickle down business when it comes to costs. They get passed down from manufacturing to insurance companies, to hospitals, and finally to patients. Then the out-of-pocket expenses for patients will decrease overall.
Meanwhile, Dr. Ayalew Tefferi and his colleagues at the Mayo Clinic, state in Petition to Lower the High Price of Cancer Drugs that “creating a post-FDA approval review” that will allow the FDA to compare any newer versions of drugs to their old ones. Currently the FDA can approve the biologics of a drug. However, they can neither “review…[nor] propose a fair price” nor can they “distinguish the amount of improvement from a new drug to its predecessor” (Tefferi). For example a marketed drug may not necessarily improve quality of life, but instead it increases life span for an individual suffering from a life-threatening disease by a matter of a few weeks. This drug costs significantly more than its predecessor while a price is again put on length of human life. And while a few weeks with a loved one are cherished, the medication neither prevents patient suffering, nor does it significantly impact the healthcare industry by qualifying as a breakthrough. These are the kinds of medications that are bankrupting our health care system. This policy changes the approval process so that prior to approval, the FDA must check medications before biomedical firms sit down with CMS and discuss price negotiations for a medication where the value of medication is far less than its cost. According to Dr. Lowell Schnipper, Chief of Harvard Medical School, and his colleagues, “there is increasing emphasis on providing clear and objective information not only on clinical benefits and risks of treatment options but also on cost” (2565). The impact of an ideal treatment plan on patients and their families yields happier consumers. The optimal image of patient satisfaction originates from more affordable prices on care and the value of care given. And as the face of healthcare is changing, so must its policies to keep the system alive and sustainable by ensuring prescription drug prices are no longer based off of bearable market value.
The bill’s coverage spans across the country from rich to poor and conservative to liberal. Everybody deserves a fighting chance at affordable medications that will improve their quality of life. According to Hagop Kantarjian, M.D., Chairman of the Leukemia Department at the University of Texas Medical Anderson Cancer Center, streamlining the drug approval process and bureaucratic burdens will “eliminate unnecessary steps that increase cost, delay-timelines, shorten patent terms” (“High Cancer Drug Prices in the United States: Reasons and Proposed Solutions”). This will benefit pharmaceutical companies as well, because they will have to pay less than what they currently do in terms of manufacturing and approval costs. And while the gross profit of biomedical companies will decrease, the percent decrease in profit will stagnate similar to the current numbers. And it is likely that even the slight decrease in gross profit for pharmaceutical companies will be a drop in the ocean to the $300 billion, international industry (WHO). The policy will turn right back and benefit consumers and insurance companies. With the help of financial analysts, it has the potential to lower insurance premiums and get drugs on the market faster. This would also promote more market competition, resulting in lowering costs of even specialty medications. H.R. Bill 3513 will support consumers by lowering governmental costs to the large biomedical corporation. And ironically the domino effect that plagues industry will turn around to still benefit everyone across the board.
Various countries have different methods of regulation upon the inflation of drug prices and how they are set, such as Great Britain’s Pharmaceutical Price Regulation Scheme of 2014. This requires their government to set a cap on all medications that enter its market. Contrastingly, United States law does not currently contain regulative policies applicable to the skyscraper prescription drug prices set by for-profit companies. Dr. Ayalew Tefferi and his colleagues depict the opposing situations of nations like Great Britain where the profit-driven market only commands 5% gross domestic product compared to the 18% demand of America’s gross domestic product. So the considerably more expensive American medications are marketed at a different rate than their European or Asian counterparts—and many times by the same company no less! Another speed bump is hit when Americans attempt to outsource medication by ordering from abroad. However, in an effort to prevent illegal substance abuse, the FDA placed a 90-day limit on imported medications as a part of the 2006 revision to the 2003 Medicare Prescription Drug, Improvement, and Modernization Act—an example of misplaced results to good intentions. Thus, preventing American physicians from prescribing and residents from consuming imported drugs. However, both the H.R. Bill 3513 and the Mayo Clinic Petition propose to amend the Federal Food, Drug, and Cosmetic Act, allowing the importation by individuals of prescription drugs from Canada and other countries, and revise the Federal Trade Commission Act, enforcing restrictions on restriction access to generic drugs (U.S. House & Tefferi). This will result in increased competition through the international market rather than the direct source of American drugs. Through negotiations of with CMS and market regulations and the modifications to the Federal Trade Commission Act and Federal Food, Drug, and Cosmetic Act, pharmaceutical firms will have to succumb to similar rates for the same medication across the world. The goal exists to diminish the exclusivity of the drug manufacturing market, as a direct response to free-market regulation of a dire commodity in the United States.
 icons at the top right corner of the subsection.
icons at the top right corner of the subsection.